General Introduction to ISO 13485
ISO 13485 is an international standard that specifies requirements for a Quality Management System (QMS) for organizations involved in manufacturing, trading, distribution, and services related to medical devices.
The standard strongly focuses on safety, product efficacy, risk control throughout the medical device lifecycle, and compliance with regulatory requirements of various markets (EU, Canada, Japan, etc.).
Properly understanding and applying ISO 13485 helps enterprises:
- Enhance product quality & safety
- Complete regulatory documentation for market authorization
- Increase credibility with customers & global partners
- Build a solid foundation for manufacturing, importing, and distributing medical devices
Objectives of ISO 13485 Awareness Training
This course is designed to help participants:
1. Understand the nature and scope of ISO 13485
- Significance of the standard in the medical device industry
- Relationship with ISO 9001 and related regulatory requirements
2. Grasp the structure and requirements of each clause
- Risk-based thinking
- Document & record management
- Design & development control
- Supplier management
- Production, traceability
- Measuring equipment control
- Post-market surveillance & incident reporting
3. Know how to apply in business practice
- Minimum requirements for establishing QMS
- Common errors in ISO 13485 assessment
- Preparation for certification assessment
4. Create foundation for system implementation or internal audit
Training Program Content
The program can be customized by sector: manufacturing — import — distribution — repair — medical device technical services.
1. General Introduction
- Overview of medical device industry and regulatory requirements
- Purpose & benefits of ISO 13485
- Scope of application
2. Systems Thinking & Risk-Based Approach
- QMS concept in medical devices
- Risk management
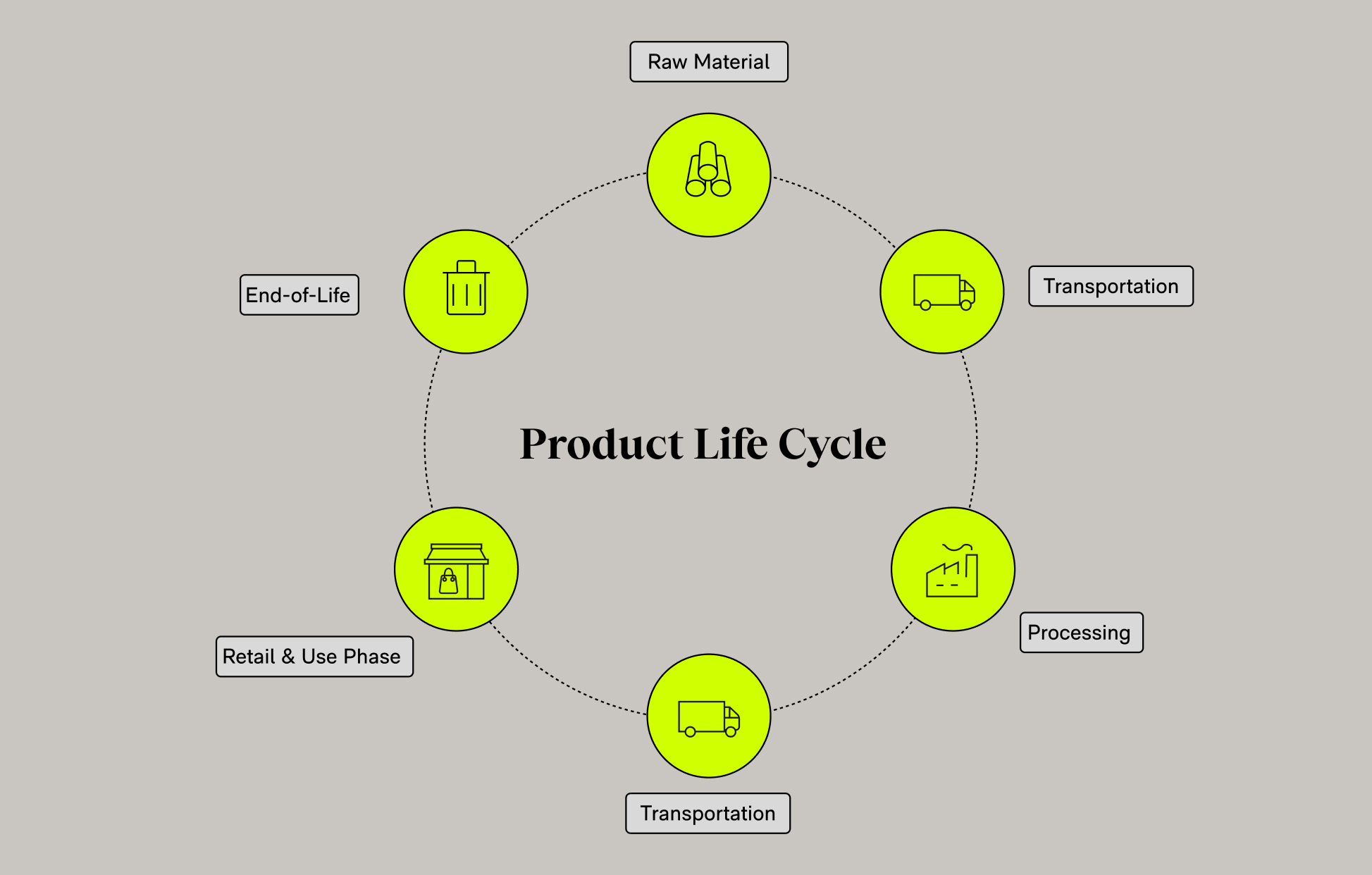
- Product lifecycle integration
3. ISO 13485:2016 Requirements by Clause Structure
- Clause 4: Quality Management System
- Clause 5: Management Responsibility
- Clause 6: Resource Management
- Clause 7: Product Realization
- Design & Development
- Purchasing & Supplier Control
- Production & Environmental Control
- Packaging, Preservation, Transportation
- Clause 8: Measurement — Analysis — Improvement
4. Documentation and Records per ISO 13485
- Required SOPs, WIs, Forms
- Version control, archiving requirements
5. Certification Assessment Preparation
- Certification process
- Analysis of common non-conformities
- Practical experience in medical device enterprises
Target Participants
- Executive Board
- QA – QC – RA – Production Heads/Deputies
- Risk management, warehouse — operations departments
- New personnel needing to understand quality systems
- Enterprises preparing for ISO 13485 certification
Training Methodology
- Lectures combined with real case studies
- Situation discussions by enterprise type
- Guidance on risk identification & basic documentation
- Direct Q&A based on participants’ business models
Benefits of Attending the Course
- Clear understanding of ISO 13485 requirements and practical application
- Enhanced operational capability, risk control, and product quality
- Optimized processes to meet regulatory inspections
- Time and cost savings in certification implementation
– – – – – – – –
Contact ISC Global Today
Register for free consultation or request a quote today to receive a preliminary assessment of energy savings potential at your enterprise.
ISC Global Co., Ltd.
Hotline: +84 933096426 – +84 868 591 260
Email:
Website:
Contact Vietnam representative: Duc Luong Services
Hotline: +84 933096426 – +84 868 591 260
Email: ducluongservices@gmail.com
Website:
STC VN Co., Ltd.
Hotline: +84 933096426 – +84 868 591 260
Email: info@staunchlyservices.com.vn
Website: https://stauchlyservices.com.vn




No responses yet